US: Very high levels of Covid in Louisiana, Tennessee, Alabama, Virginia, Georgia, North Carolina and Vermont
CDC wastewater data:https://t.co/TG7ksAzRje pic.twitter.com/SD3ncUHMe5
— CoronaHeadsUp (@CoronaHeadsUp) February 23, 2024
No longer pandemic, just endemic — with flareups.
Last night's update: 187,000 new cases, 1,569 new deaths https://t.co/U55lnuFS7Q
— BNO News (@BNOFeed) February 26, 2024
So far this year, more than 2.2 million cases of COVID-19 have been reported in the U.S., causing 175,000 hospitalizations and more than 17,000 deaths.
— BNO News (@BNOFeed) February 26, 2024
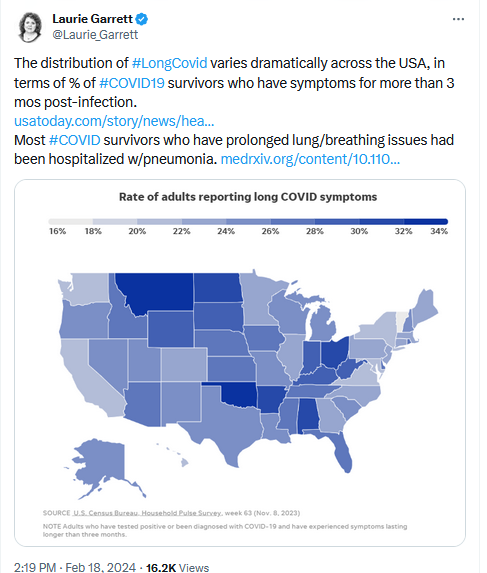
USA Today: Federal estimates suggest at least 16 million Americans have long COVID. Maybe 4 million of them are disabled by it.
The mass media finally wakes up to Long Covid. 🙄 https://t.co/V5W3noO371
— CoronaHeadsUp (@CoronaHeadsUp) February 26, 2024
======
China: Prevalence of bacteria, fungi, and virus coinfections with SARS-CoV-2 Omicron variant
82.3% severe patients coinfected with at least one additional pathogen
Other viruses 73.5%
Bacteria 58.7%
Fungi 27.1% https://t.co/pIZhI9hlrJ— CoronaHeadsUp (@CoronaHeadsUp) February 25, 2024
Japan: Number of COVID-19 and flu cases remain high
"New COVID-19 cases reported in the week to Feb. 11 stood at 13.75 per institution. By prefecture, Ishikawa had the largest number, at 21.91, followed by Aichi Prefecture, at 20.06."
Japan Timeshttps://t.co/P4ODZ8uGG1
— CoronaHeadsUp (@CoronaHeadsUp) February 24, 2024
South Australia: 'Covid rips through school staff rooms as hundreds call in sick'
The latest version of the article is here:https://t.co/BZuDgMAhV6
— CoronaHeadsUp (@CoronaHeadsUp) February 23, 2024
Sputnik V is the Russian Covid vaccine and it was tested originally on soldiers in lieu of standard human trials. If Sacks wanted to be consistent in his conspiracism, he’d ask if that vaccine is perhaps to blame for Russian soldiers’ serial fuck-ups in Ukraine. https://t.co/G4169mrgCr
— Michael Weiss (@michaeldweiss) February 27, 2024
Norway: Third largest-ever Covid wave subsides
"Many are still suffering from extreme fatigue, forgetfulness, shortness of breath and erratic heartbeats along with other ailments." https://t.co/cFJ3HBm0QM:https://t.co/WK6r54LXtY pic.twitter.com/Uj6Kb7vMyk
— CoronaHeadsUp (@CoronaHeadsUp) February 24, 2024
Sweden: Many young people suffer from brain fog after the pandemic
32% of young adults feel they have brain fog. A fifth of respondents worried about suffering from exhaustion due to brain fog.
If insurance surveyhttps://t.co/Z7ytL4cCCF
— CoronaHeadsUp (@CoronaHeadsUp) February 24, 2024
UK: High number of persistent COVID-19 infections in the general population
Researchers estimate that between one in a thousand to one in 200 of all infections may become persistent, and last for at least 60 days.
Oxford University:https://t.co/ifoZl3ybc7
— CoronaHeadsUp (@CoronaHeadsUp) February 22, 2024
Scotland: True prevalence of long-COVID in a nationwide, population cohort study
6.6% at 6 months
6.5% at 12 months
10.4% at 18 monthsNature Communicationshttps://t.co/KGN7f9ymTR
— CoronaHeadsUp (@CoronaHeadsUp) February 27, 2024
Brazil, Mato Grosso: Almost 20,000 Covid cases so far this year
62 people are hospitalized, and 48 deaths have resulted from more serious cases of the disease.https://t.co/Lgg7CHELh5
— CoronaHeadsUp (@CoronaHeadsUp) February 27, 2024
======
New analysis reveals #SARSCoV2 infection & vaccine-induced antibody responses are long-lasting. Research conducted by microbiologists at Icahn School of Medicine in NY City. Conclusions about long-term responses are based on >8k samples collected ovr 3 yrs https://t.co/TOPmqnOofT
— delthia ricks 🔬 (@DelthiaRicks) February 26, 2024
A new #SciencePerspective by @zalaly and @EricTopol identifies key issues that need to be prioritized in the study of #LongCovid.
Learn more: https://t.co/KEcm7XC1TL pic.twitter.com/sq9zfANuA2
— Science Magazine (@ScienceMagazine) February 26, 2024
Our waning of #SARSCoV2 spike-specific IgG antibodies after vaccination and/or infections is not as bad as thought, levels stabilize and are durable after 7-9 monthshttps://t.co/EGkclqzuBk @ImmunityCP @VivianaSimonLab @florian_krammer pic.twitter.com/VY2CFPMQ5J
— Eric Topol (@EricTopol) February 22, 2024
Elusive immune cells dwelling in 'hidden niches' of the bone marrow may be key to #SARSCoV2 vaccination. By @DelthiaRicks https://t.co/M3V69CTd6W… via@medical_xpress
— delthia ricks 🔬 (@DelthiaRicks) February 27, 2024
Johns Hopkins study in humanized mouse model found that long-term infection with #SARSCoV2 results in brain deposits of clumped and tangled "tau" proteins commonly associated with Alzheimer's https://t.co/Zqq8P3TEYo pic.twitter.com/WUv716RBLW
— delthia ricks 🔬 (@DelthiaRicks) February 23, 2024
Scientists pinpoint the possible underlying cause of brain fog in #LongCovid: leaky blood vessels https://t.co/QyNIV42sBf
— delthia ricks 🔬 (@DelthiaRicks) February 22, 2024
#Microbiome: Researchers are exploring whether gut microbes cause some #Covid patients to have higher blood clot risk https://t.co/qLNnhAe90k
— delthia ricks 🔬 (@DelthiaRicks) February 23, 2024
Study: Viral shedding of SARS-CoV-2 in body fluids associated with sexual activity
82.6% in saliva
1.6% in semen
2.7% in vaginal secretion
3.8% in urine
31.8% in faeces/rectal swabsH/t @ejustin46
BMJ Open:https://t.co/8v3bBGz8Q5 pic.twitter.com/ltnG5n0Nnq
— CoronaHeadsUp (@CoronaHeadsUp) February 24, 2024
Here we go again: The CDC is tracking a new #Covid variant—BA.2.87.1 https://t.co/Adx7b8GCLv
— delthia ricks 🔬 (@DelthiaRicks) February 25, 2024
Study: SARS-CoV-2 contamination on healthy individuals' hands in community settings during the COVID-19 pandemic
"SARS-CoV-2 RNA was detected in three of the 925 samples. The RT-qPCR-positive samples did not contain viable viruses."#CovidIsAirborne https://t.co/ETNrlc2j6B
— CoronaHeadsUp (@CoronaHeadsUp) February 26, 2024
======
This is good reporting on the amoral entrepreneurs who fuel not just misinformation but danger and death….
Gift link:
Tax records reveal the lucrative world of covid misinformation https://t.co/77Sjb6Moz3— Jeff (Gutenberg Parenthesis) Jarvis (@jeffjarvis) February 21, 2024
“Non-profits”, under law if not logic. Worth reading the whole article [gift link]:
… Children’s Health Defense, an anti-vaccine group founded by Robert F. Kennedy Jr., received $23.5 million in contributions, grants and other revenue in 2022 alone — eight times what it collected the year before the pandemic began — allowing it to expand its state-based lobbying operations to cover half the country. Another influential anti-vaccine group, Informed Consent Action Network, nearly quadrupled its revenue during that time to about $13.4 million in 2022, giving it the resources to finance lawsuits seeking to roll back vaccine requirements as Americans’ faith in vaccines drops.
Two other groups, Front Line Covid-19 Critical Care Alliance and America’s Frontline Doctors, went from receiving $1 million combined when they formed in 2020 to collecting more than $21 million combined when they formed in 2020 to collecting more than $21 million combined in 2022, according to the latest tax filings available for the groups.
The four groups routinely buck scientific consensus. Children’s Health Defense and Informed Consent Action Network raise doubts about the safety of vaccines despite assurances from federal regulators. “Vaccines have never been safer than they are today,” the Centers for Disease Control and Prevention said on its webpage outlining vaccine safety…
Arthur Caplan, head of the division of medical ethics at the New York University Grossman School of Medicine, said that in his view, the four groups endanger lives with their spread of misinformation.
“These groups gave jet fuel to misinformation at a crucial time in the pandemic,” Caplan said. “The richer they get, the worse off the public is because, indisputably, they’re spouting dangerous nonsense that kills people.”
The influx of pandemic cash sent executive compensation soaring, boosted public outreach, and seeded the ability to wage legislative and legal battles to weaken vaccine requirements and defend physicians accused of spreading misinformation…
Many of the contributors are not known because nonprofits are generally not required to publicly report their donors. But nonprofits are supposed to disclose groups to which they contribute more than $5,000. In addition to the tax forms filed by the four groups, The Post reviewed more than 330 filings by nonprofits that donated to the groups during the pandemic. Half of those gifts over $100,000 were made through a tax vehicle popular among the ultrawealthy known as “donor-advised funds,” which allow individuals to obscure their identities. The Post identified two funds dedicated to advancing biblical, libertarian or conservative values that each had given at least $1 million in total to at least three of the groups since 2020…
As the groups’ coffers grew, so did the salaries of some top executives. Children’s Health Defense paid Kennedy, then chairman and chief legal counsel and now an independent candidate for president, more than $510,000 in 2022, double his 2019 salary, tax records show. Informed Consent Action Network paid Executive Director Del Bigtree $284,000 in 2022, a 22 percent increase from 2019. Bigtree now works as communications director for Kennedy’s presidential campaign.
Some of the individuals behind the family foundations or trusts that fund the four groups also contributed the legal maximum in personal donations to Kennedy’s presidential bid, according to OpenSecrets, which tracks political donations…
The groups contributed to a media ecosystem that spread misinformation during the pandemic. Children’s Health Defense started an internet TV channel with daily programming casting doubt on vaccine safety, said Dorit Reiss, a professor at the University of California College of the Law at San Francisco who tracks the influence of these organizations. Informed Consent Action Network spent nearly $6 million on online “educational programs” in 2022 that the group says reached more than 6 million viewers in 209 countries, according to tax filings.
Caplan said that in his view, the four groups “were able to take advantage of fear and panic and anger at a crucial time in the pandemic and raise considerably more money to tell people what some of them wanted to hear.”…
COVID-19 Coronavirus Updates: February 28, 2024Post + Comments (35)