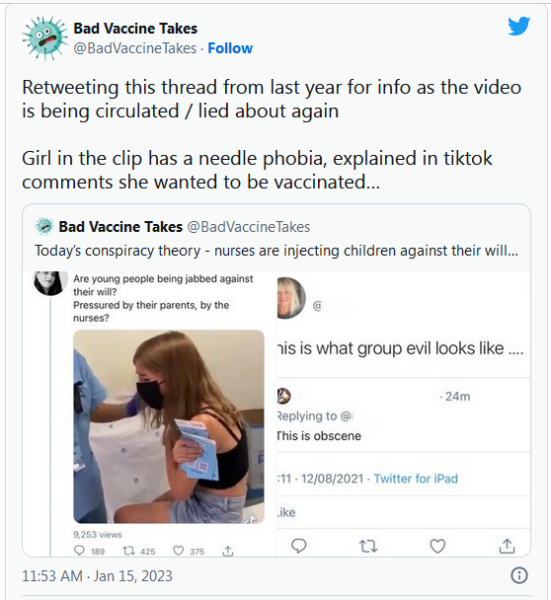
Good new rundown on bivalent booster benefithttps://t.co/mGmGjb0e3s by @SmithDanaG @NYTHealth pic.twitter.com/s4Cofcsxsk
— Eric Topol (@EricTopol) February 2, 2023
What the end of the #Covid public health emergency could mean for you. The emergency expires in May and it will complicate access to tests & treatment in the U.S. https://t.co/XFEvFb630x
— delthia ricks 🔬 (@DelthiaRicks) February 3, 2023
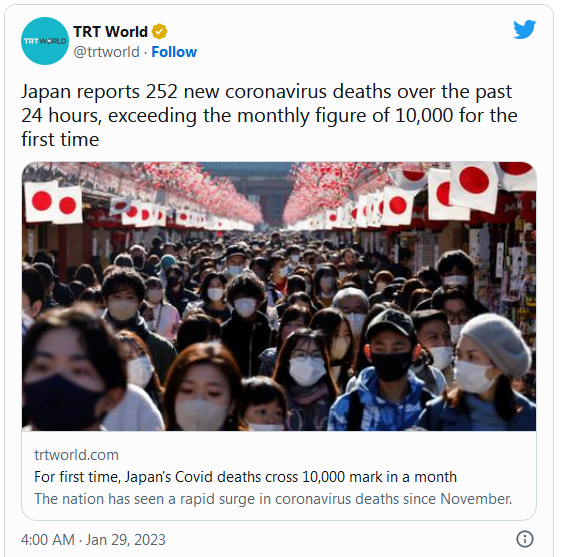
COVID-19 Coronavirus Updates: Monday / Tuesday, Jan. 30-31
President Biden will end the U.S. #Covid emergencies May 11. He told Congress the two nat'l Covid responses—the nat'l emergency & the public health emergency—will end in May. The move restructures the U.S. response to accepting Covid as an endemic threat https://t.co/mUZLGg5nWP
— delthia ricks 🔬 (@DelthiaRicks) January 31, 2023
Weekend Reading: Longer Updates On the Not-Yet-Over Pandemic
XB what? BQ huh? Do you need to keep up with Omicron’s ever-expanding offspring? https://t.co/OFq4ZwwRRy
— STAT (@statnews) January 12, 2023
It’s like clockwork now. Every few months, we’re warned that the Omicron variant of the SARS-CoV-2 virus has spawned yet another subvariant, this one even more transmissible than the ones it is fast overtaking.
The new entity is given a name, an unwieldy string of letters and numbers separated by periods. There’s discussion — some of it breathless — on Twitter and in the media about the threat the new subvariant poses. People who are still following Covid-19 news worry. People who are determined to ignore Covid pay no attention…
The cycle has some experts wondering about how useful these discussions are. We aren’t, after all, obsessing about which strain of H3N2 flu has been causing most of the illness that has cycled through the United States in this abnormally early flu season. That’s because new strains of existing flu viruses may make us more vulnerable to infection, but they don’t render us defenseless against influenza. The same is true with SARS-2 subvariants — but that sometimes gets lost in the back and forth.
“This keeps happening every couple of months. I sort of feel like it’s Groundhog Day, except with ‘scariants,’” said Angela Rasmussen, a coronavirus virologist at the University of Saskatchewan’s Vaccine and Infectious Disease Organization, using a term coined by Eric Topol.
(For the record, Topol, founder and director of the Scripps Research Translational Institute, disagrees vehemently with the notion that people don’t need to pay much attention to which variant or subvariant is currently circulating, arguing among other things that the public discussion could encourage more people to get the latest booster shots.)…
Weekend Reading: Longer Updates On the Not-Yet-Over PandemicPost + Comments (46)
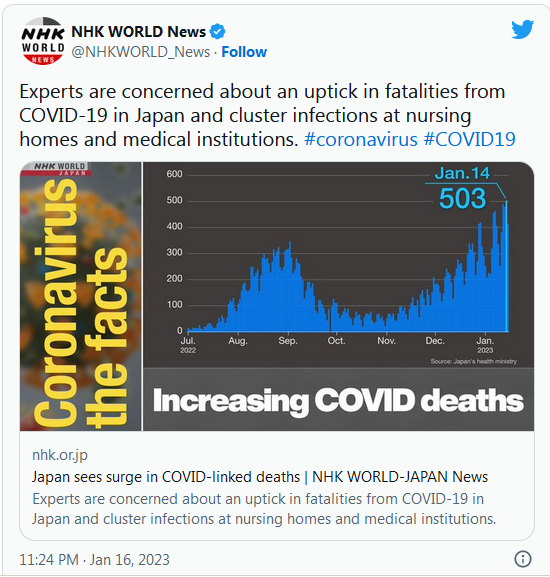
COVID-19 Coronavirus Updates: Thursday / Friday, Jan. 26-27
Advisers to the U.S. Food and Drug Administration on Thursday unanimously voted in favor of using the same coronavirus strain for the initial COVID-19 vaccine doses and the boosters, to simplify the vaccination regimen in the United States. https://t.co/Hrxq2VlTpd
— Reuters Health (@Reuters_Health) January 27, 2023
COVID-19 Coronavirus Updates: Thursday / Friday, Jan. 26-27Post + Comments (66)
COVID-19 Coronavirus Updates: Monday / Tuesday, Jan. 23-24
I got my last COVID shot and my last flu shot on the same day, one in each arm. Poke, poke, done. I don't have any objection to doing that in perpetuity.https://t.co/BbfH080pSf
— John Scalzi (@scalzi) January 23, 2023
Gift (unpaywalled) link:
… The agency, in briefing documents released in advance of a meeting this week with its vaccine advisers, said the goal is to determine in the spring which strain will pose the greatest threat the following winter. A vaccine targeting that strain would then be administered in the fall, in the hopes that a streamlined effort would encourage vaccine uptake.
The evidence suggests that “moving forward, most individuals may only need to receive one dose” of a coronavirus vaccine “to restore protective immunity for a period of time,” the FDA said in the briefing papers. The change in strategy is expected to be one of the key topics discussed at the agency’s vaccine advisory committee meeting scheduled for Thursday. The advisers’ views will be taken into consideration when the FDA decides on a final plan.
The FDA’s proposal represents a shift from its strategy of pushing boosters to counter variant threats — an effort that ran into resistance from the public, which showed little interest in getting repeated shots. The new blueprint for an annual dose of protection drew conflicting reactions…
The FDA suggested Monday that the annual formulation for coronavirus vaccines would be chosen every June, in time to be manufactured and administered by September as part of a yearly immunization campaign. The goal would be to select the strain most likely to be dominant in the winter, when people are indoors and coronavirus cases typically rise. The strategy suggests a recognition that the virus has become endemic, a part of the American health-care scene that will recur year after year.
If there were an emergency — the appearance of a more dangerous variant capable of evading vaccine-endowed immunity — the FDA would call an impromptu meeting of its vaccine advisers and select a new strain to counter the threat, the agency indicated.
While most people would get an annual shot, people who are older, very young or immunocompromised, or who have serious health problems, might need two doses, the FDA briefing document said.…
The FDA, according to the document, also is seeking an endorsement from the committee to retire the original vaccination formula that entered the market in late 2020 — the monovalent version that targets the original virus. That means the updated, bivalent booster would be used for the original two-shot series as well as for the booster — until a new strain is chosen…
COVID-19 Coronavirus Updates: Thursday / Friday, Jan. 19-20
#COVID19 #VariantDashboard – #UnitedStates
BQ.1.1 vs XBB.1.5: Tug of War!
TOP lineages (#15DAYTRENDS):
21.9% BQ.1.1
21.7% XBB.1.5
6.3% BQ.1
3.2% XBB.1
2.5% BQ.1.2
2.4% BQ.1.1.5
2.4% BQ.1.3Tracker: https://t.co/C4MNBUx4a2
Updated: 01/17/20231/n pic.twitter.com/HpRa3IcvCd
— Raj Rajnarayanan (@RajlabN) January 18, 2023
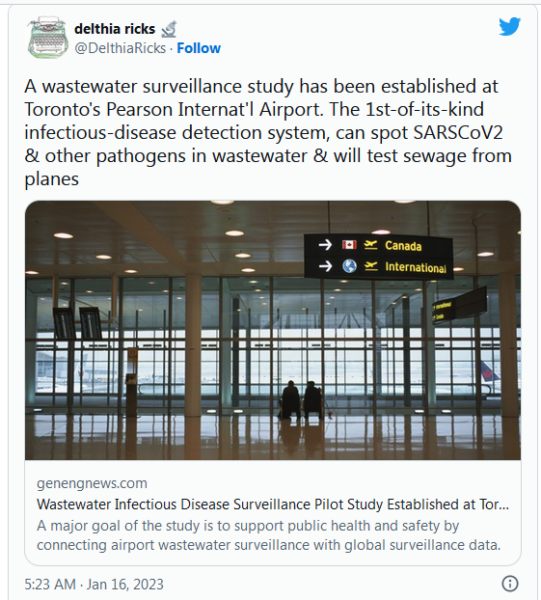
COVID-19 Coronavirus Updates: Monday / Tuesday, Jan. 16 / 17
… As chief science officer for the administration’s Covid-19 response, Dr. Kessler, 71, has operated largely unseen by the public. But his work — on issues like setting up mass vaccination sites, pushing for the development of antiviral medicines and distributing reformulated booster shots — has affected the lives of millions of Americans.
“Six hundred sixty-five million vaccines, 13 million antivirals,” Dr. Kessler said in a brief interview on Friday, referring to the number of doses that Americans have taken since December 2020, shortly before President Biden took office. “We did what we set out to do.”…
Dr. Kessler’s departure signals the end of Operation Warp Speed, which was started by the Trump administration to develop and distribute coronavirus vaccines. Although the Biden administration dropped the name, the mission of the program under Dr. Kessler remained the same. Now the Biden administration is working to shift Covid vaccination from a government-run effort to one that will be handled by the private sector.
In his role, Dr. Kessler was responsible for negotiating with drug companies to make certain that vaccines were available to anyone who wanted one — at a price for the government that was far lower than what companies want to charge on the commercial market in the future.
It seems unlikely that the Biden administration will replace Dr. Kessler. Officials say the Department of Health and Human Services has other doctors, scientists and public health officials with expertise who will work with the White House and private industry on vaccine research and development.
Dr. Kessler had hopes of building a more permanent infrastructure for vaccine development and manufacturing. In late 2021, the administration announced a plan, drafted by Dr. Kessler, to invest billions of dollars to expand vaccine manufacturing capacity by partnering with industry to prepare for future pandemics…
The plan was set aside, however, when Congress refused to give the administration any additional money to spend on vaccines. The billions the administration had hoped to spend to ramp up vaccine manufacturing capacity went instead to purchasing the updated booster shots now being distributed…
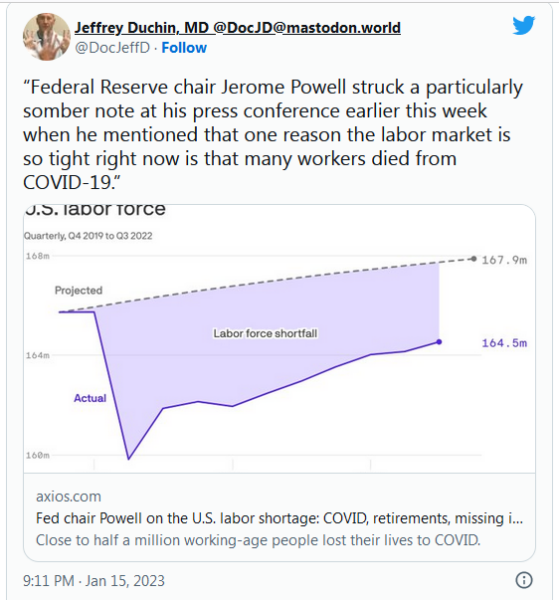
After +1M people in the US died in the Covid pandemic, you might have thought there'd be an after-action review of the country's response & traction for changes so next time goes better. But you'd have been wrong. @rachelcohrs explores where things stand. https://t.co/rGhJsCswYP
— Helen Branswell ???? (@HelenBranswell) January 11, 2023